Molecular Imaging
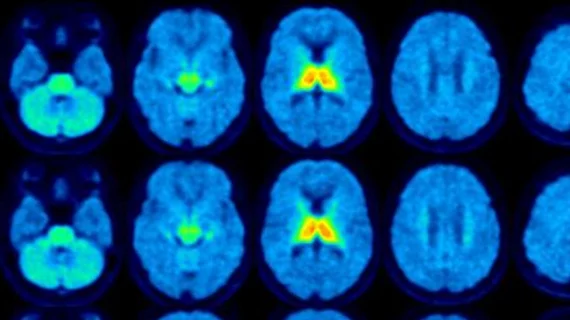
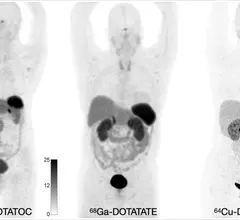
Molecular imaging (also called nuclear medicine or nuclear imaging) can image the function of cells inside the body at the molecular level. This includes the imaging modalities of positron emission computed tomography (PET) and single photon emission computed tomography (SPECT) imaging. How does PET and SPECT imaging work? Small amounts of radioactive material (radiopharmaceuticals) injected into a patient. These can use sugars or chemical traits to bond to specific cells. The radioactive material is taken up by cells that consume the sugars. The radiation emitted from inside the body is detected by photon detectors outside the body. Computers take the data to assemble images of the radiation emissions. Nuclear images may appear fuzzy or ghostly rather than the sharper resolution from MRI and CT. But, it provides metabolic information at a cellular level, showing if there are defects in the function of the heart, areas of very high metabolic activity associated with cancer cells, or areas of inflammation, data not available from other modalities. These noninvasive imaging exams are used to diagnose cancer, heart disease, Alzheimer’s and Parkinson’s disease, bone disorders and other disorders.