By using focused ultrasound, clinicians can open parts of the blood-brain barrier to administer new treatments for Alzheimer’s disease, according to a pilot study presented at the Radiological Society of North America’ annual meeting.
Alzheimer’s disease remains the most common cause of dementia. And while there is no known effective treatment, bypassing the blood-brain barrier—a network of blood vessels and tissue protecting the brain—enabled researchers to administer targeted drug and stem-cell treatments to multiple patients.
"The results are promising," co-author Rashi Mehta, MD, associate professor at West Virginia University, said in an RSNA announcement. "We were able to open the blood-brain barrier in a very precise manner and document closure of the barrier within 24 hours. The technique was reproduced successfully in the patients, with no adverse effects."
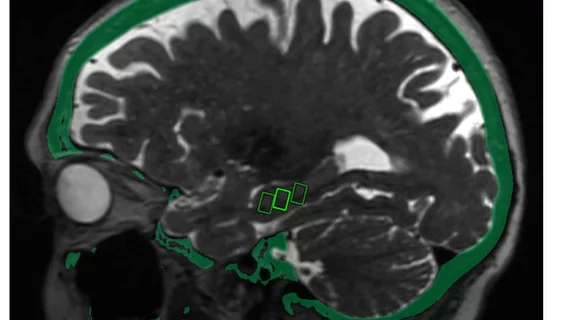
For their study, Mehta and colleagues administered low-intensity focused ultrasound (LIFU) to three women, ages 61, 72 and 73, targeting specific sites in the brain associated with memory. The patients, all with early stage Alzheimer’s and amyloid plaques linked to the disease, received three consecutive treatments at two-week intervals.
Each patient underwent brain MRIs after treatment, which researchers used to confirm that the blood-brain area opened within their intended target areas. That barrier closed again in each patient within 24 hours.
Delivering MRI-guided LIFU requires a patient to wear a helmet equipped with more than 1,000 separate ultrasound transducers that deliver sound waves targeted at specific brain areas. Patients also receive an injection of contrast agent in the form of microscopic bubbles which, when hit by ultrasound, change shape and size, loosening the blood-brain barrier.
Opening this protective layer has resulted in positive effects within animals, which may be due to an increased flow of fluid that rids the brain of toxic substances. LIFU could enable this cleansing and help deliver therapeutic drugs into the brain of those suffering from Alzheimer’s.
Going forward, Mehta and her colleagues hope to expand LIFU to an even wider group of subjects.
"We'd like to treat more patients and study the long-term effects to see if there are improvements in memory and symptoms associated with Alzheimer's disease," Mehta added. "As safety is further clarified, the next step would be to use this approach to help deliver clinical drugs."