A team of researchers from the University of Illinois at Urbana-Champaign has developed a portable optical imaging system that can visualize molecular features of breast tissue after it's been surgically removed from a patient, according to research published online Dec. 19 in Science Advances.
Able to track tumor progression, the tool may provide cancer researchers more in-depth information regarding cancer tissue pathology and diagnostics in real-time.
"We believe that capturing the dynamic cellular and molecular features in freshly removed or biopsied tissue specimens contains valuable diagnostic and prognostic information that is currently lost when specimens are placed in a fixative and essentially killed quickly in order to preserve structure," lead researcher Stephen Boppart, PhD, professor in electrical and computer engineering at the University of Illinois at Urbana-Champaign, said in a prepared statement. "Our imaging platform and methodology allow us to extract this new information in real-time, at the point-of-procedure."
The researchers' portable optical imaging system uses precise light pulses to image tissue in four modalities at the same time, allowing the team to study simultaneous processes within cells and tissue that make up the breast tumor.
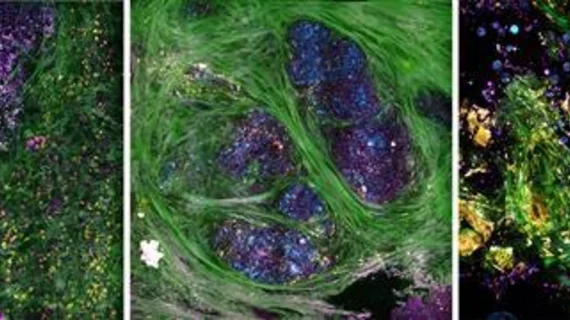
Collagen fibers appear in green; elastin fibers and flavin adenine dinucleotide-containing cell cytoplasm appear in yellow; cell membranes, lipid boundaries, and extracellular vesicles (EVs) appear in magenta; and nicotinamide adenine dinucleotide in the cells and lipids appears in cyan, according to the researchers.
For their study, the team used their imaging tool during breast cancer surgeries to image cancerous tissue within 30 minutes of breast tumors being extracted from patients. The researchers then identify molecular signatures inside the tissue which were associated with metabolic activity within individual cells making up the tumor’s microenvironment.
Tumor-related EVs were also measured, which play an essential role in cancer progression and may be developed as a potential biomarker for future cancer diagnoses.
With their technology, the researchers found that cancerous tissue exhibited increased EV densities and had shorter tumor-to-margin distance than healthy breast tissue.
In the future, they hope to use the imaging system on tissue specimens obtained from needle-biopsy procedures routinely performed in outpatient settings and samples from the operating room.
“These results suggest the feasibility and future potential for implementing intraoperative label-free nonlinear optical imaging to investigate the human breast tumor microenvironment and the spatial EV distribution, both to improve our fundamental understanding of carcinogenesis and to potentially provide new biomarkers for tumor invasiveness,” Boppart et al. wrote.